Gain a comprehensive understanding of the five states of matter through our concise notes, illustrative examples, and essential key points, enhancing your knowledge of this fundamental topic in science.
Understanding the Matter
- Imagine you’re having a delicious samosa with your friend. You can see it, touch it, and even take a bite (yum!). That samosa, everything you can interact with in this way, is what we call matter in chemistry.
- Think of matter as the solid, liquid, or gas that makes up everything around us, from the sizzling samosa to the cool breeze brushing against your face. It’s the stuff that fills the universe, from the tiniest grain of sand to the giant planets in the sky.
- Here’s the cool part: all this matter, no matter how different it seems, is made up of tiny building blocks called atoms.
- Imagine the samosa as a bunch of Lego bricks, and each brick is an atom. These atoms come together in different ways to form all the amazing things we see around us!
Now, two main things define matter:
- It has mass: This means it has weight, even if it’s super tiny. Even that sneaky mosquito buzzing around has a tiny bit of mass!
- It takes up space: No matter how hard you squish it, matter occupies a certain amount of space. Try squishing that samosa – it might get flatter, but it will still take up some space on your plate, right?
So, remember:
- Matter is all the stuff around us that we can interact with.
- It’s made up of tiny building blocks called atoms.
- It has mass and takes up space.
Think of it like your trusty samosa – it’s there, it has weight, and it takes up space on your plate (until you devour it, of course!). Understanding matter is like understanding the lego bricks of the universe, and it’s the foundation for so many things in chemistry.
What is Matter in Chemistry?
- The “matter” in chemistry is Anything that occupies space and has mass.
- This simple definition encompasses everything from the tiniest atom to the vast expanses of galaxies. While seemingly straightforward, it captures several key aspects of matter:
Occupies Space
- Matter takes up volume, meaning it excludes other matter from occupying the same space at the same time. You can’t, for example, fit your hand through a solid wall without displacing some of the wall material.
Has Mass
- Matter possesses inertia, meaning it resists changes in its state of motion. It takes a force to get something moving, and another force to stop it.
This definition also implies two important distinctions:
Matter vs. energy
- Energy, unlike matter, does not have its own independent existence. It exists in relation to matter (e.g., kinetic energy of a moving object, potential energy stored in a chemical bond).
Matter vs. fields
- Fields like the electromagnetic field or the gravitational field are not considered matter as they do not themselves occupy space and have no intrinsic mass.
This definition forms the foundation for all of chemistry, as it describes the basic building blocks and properties of the universe we study. Understanding what matter is, how it interacts, and how it changes is crucial for any exploration of chemical phenomena.
It has been observed that matter exists in nature in different forms. Some substances are rigid and have a fixed shape like wood and stone; some substances can flow and take the shape of their container like water, while there are forms of matter that do not have definite shapes or sizes such as air.
Common States of Matter
Matter, the substance that makes up everything around us, can be categorized based on its physical properties and the different states it can exist in. These categories are known as states of matter.
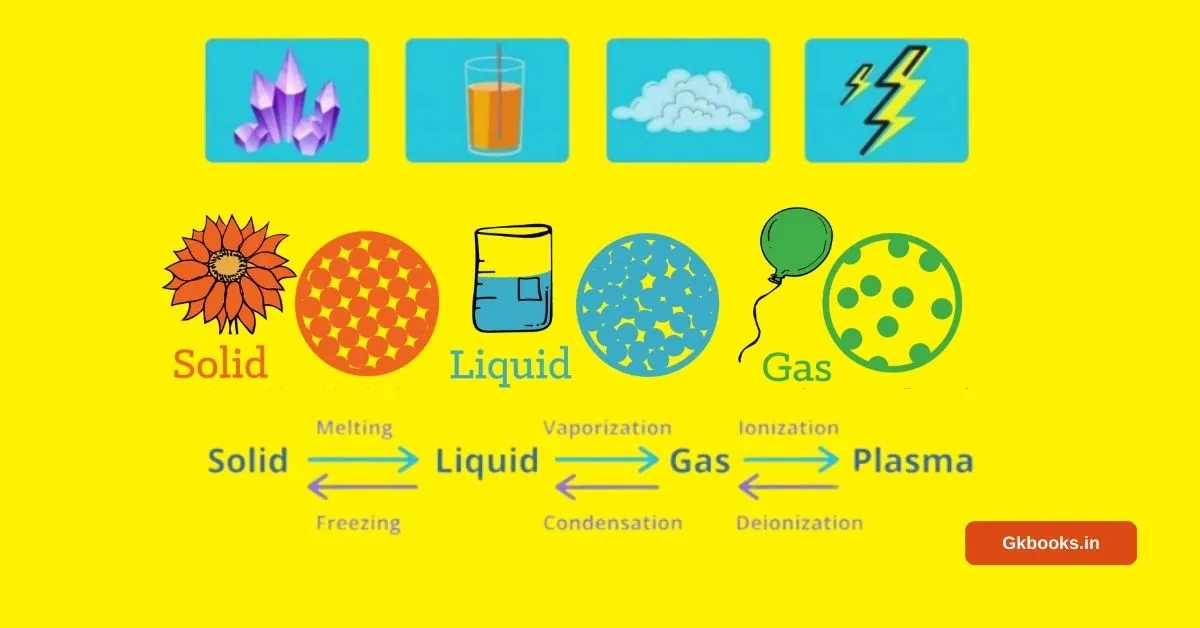
- Solid: In this state, particles are closely packed together, giving the substance a rigid form. Think of ice or a wooden desk.
- Liquid: Here, particles have more freedom to move, allowing the substance to flow. Water is a classic example of a liquid.
- Gas: In the gaseous state, particles are widely spaced and move freely. Air is a familiar gas.
Less Familiar States of Matter
Apart from solids, liquids, and gases, two lesser-known states of matter aren’t part of our everyday experience.
- Plasma: This unique state, found in stars and lightning, involves charged particles with high energy.
- Bose-Einstein Condensate: Formed at extremely low temperatures, this state exhibits quantum phenomena not seen in our daily lives.
Factors Influencing States
- Changes in matter, particularly shifts between states, are influenced by external factors like pressure and temperature.
Examples:
Consider the familiar example of ice. As its temperature rises, a significant change occurs at 0 °C (32 °F). Instead of a gradual temperature increase, the energy flows into a phase transition, marking a shift in state rather than a simple rise in temperature.

Solid States of Matter
- In solids, constituent particles such as ions, atoms, or molecules are densely packed together, with forces between them preventing free movement, allowing only vibrational motion.
- This confinement results in solids having a stable, definite shape, and a fixed volume. Altering their shape requires an external force, like breaking or cutting.
- Crystalline solids exhibit particles arranged in a regularly ordered, repeating pattern. Various crystal structures exist, with a substance potentially having more than one structure or solid phase. For instance, iron displays a body-centred cubic structure below 912 °C and a face-centred cubic structure between 912 and 1,394 °C.
- Some substances, like ice, can have multiple crystal structures or solid phases at different temperatures and pressures.
Note: The following content describes non-crystalline states of matter.
- Glasses and other non-crystalline, amorphous solids lack long-range order and are not thermal equilibrium ground states, categorizing them as nonclassical states of matter.
Phase Transitions:
- Solids can transition into liquids through melting, while liquids can solidify through freezing.
- Sublimation is the process where solids directly transform into gases, and deposition allows gases to change directly into solids.
Examples of Solids
- Solid Ice: The frozen form of water, maintaining a solid state at low temperatures.
- Sugar: Crystalline sugar represents a common example of a solid substance.
- Rock: Geological formations often exist in a solid state, exemplified by rocks.
- Wood: The sturdy and rigid nature of wood places it in the category of solids.
Liquid States of Matter
- A liquid is a nearly incompressible fluid that takes the shape of its container while maintaining a nearly constant volume, irrespective of pressure.
- The volume remains constant when temperature and pressure are consistent. When a solid is heated beyond its melting point, it transforms into a liquid, provided the pressure exceeds the substance’s triple point.
- Intermolecular (or interatomic or interionic) forces remain significant in liquids, but molecules possess sufficient energy to move relative to each other, resulting in a mobile structure.
- Unlike solids, the shape of a liquid is not fixed but is determined by its container. Generally, the volume is greater than that of the corresponding solid, with water (H2O) being a notable exception.
Temperature Considerations:
- The critical temperature denotes the highest temperature at which a particular liquid can exist.
Examples of Liquid States
- Water: A universal example, water demonstrates the fluid nature of liquids.
- Milk: The liquid form of milk exemplifies the adaptable properties of liquids.
- Blood: In the human body, blood flows as a crucial liquid, showcasing dynamic properties.
- Coffee: The beloved beverage, coffee, is a familiar example of a liquid with variable shapes.
Gaseous States of Matter
- A gas is a compressible fluid that not only takes the shape of its container but also expands to fill it.
- In a gas, molecules possess sufficient kinetic energy, minimizing the impact of intermolecular forces, especially for an ideal gas. The typical distance between neighboring molecules is significantly greater than the molecular size.
- Gases lack a definite shape or volume, occupying the entire container in which they are confined. The conversion of a liquid to a gas can occur by heating at constant pressure to the boiling point or by reducing the pressure at a constant temperature.
Vapor State and Equilibrium
- Below its critical temperature, a gas is referred to as a vapor. Compression alone, without cooling, can liquefy a vapor.
- A vapor can exist in equilibrium with a liquid (or solid), with the gas pressure equaling the vapor pressure of the liquid (or solid).
Supercritical Fluids
- A supercritical fluid (SCF) is a gas with a temperature and pressure above the critical temperature and critical pressure, erasing the distinction between liquid and gas.
- Despite having the physical properties of a gas, a supercritical fluid’s high density imparts solvent properties in some cases, leading to practical applications.
- An example is supercritical carbon dioxide, utilized in extracting caffeine during the production of decaffeinated coffee.
Examples of Gases
- Air: The mixture of gases we breathe, showcasing the diverse composition of the atmosphere.
- Helium: Known for its use in balloons, helium exemplifies the light and buoyant properties of gases.
- Nitrogen: Constituting a significant portion of the Earth’s atmosphere, nitrogen exists as a vital gas.
- Oxygen: Essential for supporting combustion and sustaining life, oxygen is a critical gaseous element.
- Carbon Dioxide: A byproduct of respiration and combustion, carbon dioxide represents a common gas in the environment.
Plasma
Definition
- Plasma, derived from the Ancient Greek word πλάσμα (plásma) meaning ‘moldable substance,’ is a fundamental state of matter.
- Characterized by a notable presence of charged particles, including ions or electrons, distinguishing it as one of the four fundamental states of matter.
Abundance and Occurrence
- Predominantly found in the universe as the most abundant form of ordinary matter.
- Commonly observed in celestial bodies such as stars (including the Sun), and it dominates the rarefied intracluster medium and intergalactic medium.
Artificial Generation
- Plasma can be artificially created by methods like heating a neutral gas or exposing it to a strong electromagnetic field.
- This artificial generation allows for controlled manipulation and application in various technologies.
Electrical Conductivity and Dynamics
- The presence of charged particles makes plasma electrically conductive.
- Individual particle dynamics and macroscopic plasma motion are influenced by collective electromagnetic fields and are highly responsive to externally applied fields.
Technological Applications
- The response of plasma to electromagnetic fields finds applications in modern devices and technologies.
- Examples include plasma televisions and plasma etching techniques.
Partially Ionized Plasma
- Depending on temperature and density, plasma may contain a certain number of neutral particles.
- When neutral particles coexist, the plasma is termed partially ionized, illustrated by phenomena like neon signs and lightning.
Transition to Plasma
- Unlike phase transitions between other states of matter, the transition to plasma is not precisely defined.
- Whether a substance is considered plasma depends on the degree of ionization, a matter subject to interpretation and context in specific phenomena.
Bose-Einstein Condensates
- A Bose-Einstein Condensate, often termed the ‘fifth state of matter,’ emerges when particles known as bosons undergo cooling to near absolute zero (-273.15 degrees Celsius or -460 degrees Fahrenheit).
- The extremely low temperatures inhibit the particles from achieving the energy required for them to assume positions that could result in interference with their distinct quantum characteristics.
- Due to the lack of energy differences that would differentiate particles, the entire group adopts a unified quantum identity.
- This collective behavior transforms the ensemble into a singular ‘super-particle’ cloud, governed by its own set of rules and operating as a cohesive unit.
States of Matter Question and Answer
Q1: What are the three common states of matter?
Answer: The three common states of matter are solid, liquid, and gas. They are made up of tiny particles with specific mass and volume. Atoms in these states have a strength of attraction between them.
Q2: Can matter be created?
Answer: According to the first law of thermodynamics, matter cannot be created or destroyed. However, energy in a closed system can be transformed from one form to another.
Q3: Is matter created or destroyed?
Answer: The Mass Conservation Law, discovered by Antoine Lavoisier in 1785, states that matter is not created or destroyed. The total mass and energy in the universe remain constant.
Q4: Is matter-energy?
Answer: Yes, matter and energy are interconnected. According to Einstein’s theory of relativity, they are fully interchangeable. They are different facets of the same thing – energy, matter, space, and time.
Q5: What is Einstein’s theory of relativity?
Answer: In 1905, Albert Einstein proposed the theory of special relativity. It states that the laws of physics are the same for all non-accelerating observers, and the speed of light in a vacuum is constant, independent of the observer’s movement.
Q10: Why is understanding matter essential in chemistry?
Understanding matter is crucial in chemistry as it forms the foundation for exploring the basic building blocks, properties, and transformations in the universe.
Q8: How was the first Bose-Einstein condensate created?
In 1995, scientists Carl Weiman and Eric Cornell used magnets and lasers to cool a sample of rubidium to a few degrees above absolute zero, creating the first Bose-Einstein condensate.
Q9: What distinguishes matter from energy?
Matter has mass and occupies space, while energy does not have independent existence and is related to matter (e.g., kinetic energy) but lacks mass.
FAQs About Matter in Chemistry
Matter in chemistry refers to anything that occupies space and has mass. It includes solids, liquids, gases, plasma, and Bose-Einstein condensates, which are the building blocks of the universe.
Matter is classified into different states: solids, liquids, and gases, based on its physical properties and the arrangement of particles. Additionally, there are less common states like plasma and Bose-Einstein condensates.
Solids have particles closely packed, a fixed shape and volume, limited particle movement, strong intermolecular forces, and low diffusion rate, examples include ice, sugar, rock, and wood.
Liquids have particles with more freedom to move, take the shape of their container, resist compression, lack a fixed shape, and exhibit a higher diffusion rate. Examples include water, milk, blood, and coffee.
Gases have widely spaced particles, negligible particle attraction, no fixed shape or volume, high compressibility, rapid diffusion rate, and elevated kinetic energy. Examples include air, helium, nitrogen, and oxygen.
Plasma consists of particles with high kinetic energy and is often created using electricity. It is observed in stars and exhibits properties like superfluidity, and flowing without friction.
BEC is a state of matter achieved at extremely low temperatures. It involves superheated particles forming a collective super-atom. BECs exhibit unique properties like slowing down light and behaving as a superfluid.
More Related Topics on Chemistry